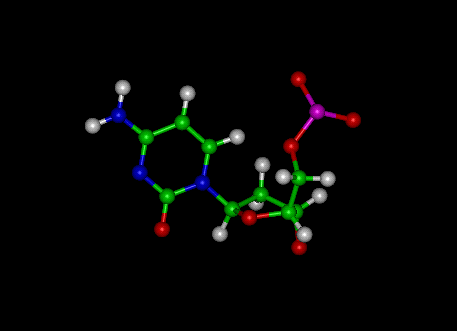
Cytosine is one of the 5 main nitrogenous bases used in storing and transporting genetic information within a cell. It is a pyrimidine derivative, with a heterocyclic aromatic ring and two substituents attached (an amine group at position 4 and a keto group at position 2). The nucleoside of cytosine is cytidine.
The other names for cytosine are 2-oxy-4-aminopyrimidine and 4-amino-2(1H)-pyrimidinone. It has a chemical formula of C4H5N3O and a molecular weight of 111.10 atomic mass units.
 |
Cytosine was first discovered in 1894 when it was isolated from calf thymus tissues. A structure was proposed in 1903, and was synthesized (and thus confirmed) in the laboratory in the same year.
Cytosine can be found as part of DNA, RNA or as a part of a nucleotide. As cytosine triphosphate (CTP), it can act as a co-factor to enzymes, and can transfer a phosphate to convert adenosine diphosphate (ADP) to adenosine triphosphate (ATP).
In DNA and RNA, cytosine is paired with guanine. However, it is inherently unstable, and can change into uracil (spontaneous deamination). Cytosine can also be methylated into 5-methylcytosine by an enzyme called DNA methyltransferase.