IP-6 Molecule
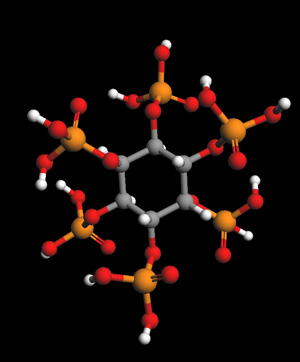
IP-6 -- Space Fill Model
To View the IP-6 Molecule in 3D using Jsmol
Inositol hexakisphosphate (IP6), or phytate when in salt form) is the principal storage form of phosphorus in many plant tissues, especially bran and seeds. Phytate is not digestable to humans or non-ruminant animals, however, so it is not a source of either inositol or phosphate if eaten directly. Morever, it chelates and thus makes unabsorbable certain important minor minerals such as zinc and iron, and to a lesser extent, also macro minerals such as calcium and magnesium.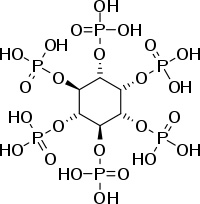
Chemical Formula for Ip-6: C6H18O24P6
Binding to Minerals
Phytic acid has a strong binding affinity to important minerals such as calcium, magnesium, iron, and zinc. When a mineral binds to phytic acid, it becomes insoluble, precipitates and will be nonabsorbable in the intestines. This process can therefore contribute to mineral deficiencies in people whose diets rely on these foods for their mineral intake, such as those in developing countries.Contrary to that, one study correlated decreased osteoporosis risk with phytic acid consumption.[12] It also acts as an acid, chelating the vitamin niacin, the deficiency of which is known as pellagra.[13] In this regard, it is an anti-nutrient, despite its possible therapeutic effects (see below). For people with a particularly low intake of essential minerals, especially those in developing countries, this effect can be undesirable. Binding of calcium with phytic acid depends on pH.
Therapuetic Effects
"...For a long time IP(6) has been recognized as a natural antioxidant. Recently IP(6) has received much attention for its role in cancer prevention and control of experimental tumor growth, progression, and metastasis. In addition, IP(6) possesses other significant benefits for human health, such as the ability to enhance immune system, prevent pathological calcification and kidney stone formation, lower elevated serum cholesterol, and reduce pathological platelet activity..." See Protection against cancer by dietary IP6 and inositol.