Water Molecule -- Chemical and Physical Properties
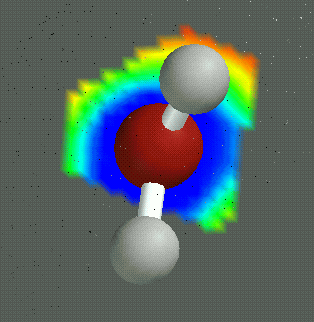
Water is a chemical compound and polar molecule, which is liquid at standard temperature and pressure. It has the chemical formula H2O, meaning that one molecule of water is composed of two hydrogen atoms and one oxygen atom. Water is found almost everywhere on earth and is required by all known life. About 70% of the Earth's surface is covered by water. Water is known to exist, in ice form, on several other bodies in the solar system and beyond, and proof that it exists (or did exist) in liquid form anywhere besides Earth would be strong evidence of extraterrestrial life.
General
The solid state of water is known as ice; the gaseous state is known as water vapor (or steam). The units of temperature (formerly the degree Celsius and now the Kelvin) are defined in terms of the triple point of water, 273.16 K (0.01 °C) and 611.2 Pa, the temperature and pressure at which solid, liquid, and gaseous water coexist in equilibrium. Water exhibits some very strange behaviors, including the formation of states such as vitreous ice, a noncrystalline (glassy), solid state of water.
At temperatures greater than 647 K and pressures greater than 22.064 MPa, a collection of water molecules assumes a supercritical condition, in which liquid-like clusters float within a vapor-like phase.
The liquid water path is a measure of the amount of liquid water in an air column.
The dipolar nature of the water molecule
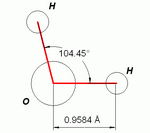
An important feature of the water molecule is its polar nature. The water molecule forms an angle, with hydrogen atoms at the tips and oxygen at the vertex. Since oxygen has a higher electronegativity than hydrogen, the side of the molecule with the oxygen atom has a partial negative charge. A molecule with such a charge difference is called a dipole. The charge differences cause water molecules to be attracted to each other (the relatively positive areas being attracted to the relatively negative areas) and to other polar molecules. This attraction is known as hydrogen bonding.
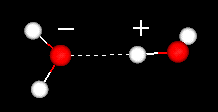 hydrogen bond between two water molecules
hydrogen bond between two water molecules
This relatively weak (relative to the covalent bonds within the water molecule itself) attraction results in physical properties such as a relatively high boiling point, because a lot of heat energy is necessary to break the hydrogen bonds between molecules. For example, sulfur is the element below oxygen in the periodic table, and its equivalent compound, hydrogen sulfide (H2S) does not have hydrogen bonds, and though it has twice the molecular weight of water, it is a gas at room temperature. The extra bonding between water molecules also gives liquid water a large specific heat capacity.
Hydrogen bonding also gives water molecules an unusual behaviour when freezing. Just like most other materials, the liquid becomes denser with lowering temperature. However, unlike most other materials, when cooled to near freezing point, the presence of hydrogen bonds means that the molecules, as they rearrange to minimise their energy, form a structure that is actually of lower density: hence the solid form, ice, will float in water. In other words, water expands as it freezes (most other materials shrink on solidification). Liquid water reaches its highest density at a temperature of 4 °C. This has an interesting consequence for water life in winter. Water chilled at the surface becomes denser and sinks, forming convection currents that cool the whole water body, but when the temperature of the lake water reaches 4°C, water on the surface, as it chills further, becomes less dense, and stays as a surface layer which eventually forms ice. Since downward convection of colder water is blocked by the density change, any large body of water frozen in winter will have the bulk of its water still liquid at 4°C beneath the icy surface, allowing fish to survive. This is one of the principal examples of finely-tuned physical properties that support life on Earth that is used as an argument for the anthropic principle.
Another consequence is that ice will melt if sufficient pressure is applied.
Structure of Water and Ice
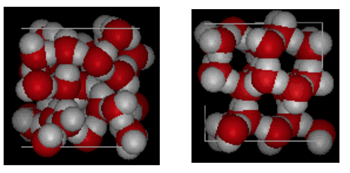
Shown above is a side by side comparison of a box 10 Angstroms across. It clearly shows that ice takes up more space because of the hydrogen bonding that occurs when the state changes from liquid to solid. In ice Ih, each water forms four hydrogen bonds with O---O distances of 2.76 Angstroms to the nearest oxygen neighbor. Because of ordered structure in ice there are less H20 molecules in a given space of volume.
Water as a solvent
Water is also a good solvent due to its polarity. The solvent properties of water are vital in biology, because many biochemical reactions take place only within aqueous solutions (e.g., reactions in the cytoplasm and blood). In addition, water is used to transport biological molecules.
When an ionic or polar compound enters water, it is surrounded by water molecules. The relatively small size of water molecules typically allows many water molecules to surround one molecule of solute. The partially negative dipoles of the water are attracted to positively charged components of the solute, and vice versa for the positive dipoles.
In general, ionic and polar substances such as acids, alcohols, and salts are easily soluble in water, and nonpolar substances such as fats and oils are not. Nonpolar molecules stay together in water because it is energetically more favorable for the water molecules to hydrogen bond to each other than to engage in van der Waals interactions with nonpolar molecules.
An example of an ionic solute is table salt; the sodium chloride, NaCl, separates into Na+ cations and Cl- anions, each being surrounded by water molecules. The ions are then easily transported away from their crystalline lattice into solution. An example of a nonionic solute is table sugar. The water dipoles hydrogen bond to the dipolar regions of the sugar molecule and allow it to be carried away into solution.
Cohesion and surface tension
The strong hydrogen bonds give water a high cohesiveness and, consequently, surface tension. This is evident when small quantities of water are put onto a nonsoluble surface and the water stays together as drops. This feature is important when water is carried through xylem up stems in plants; the strong intermolecular attractions hold the water column together, and prevent tension caused by transpiration pull. Other liquids with lower surface tension would have a higher tendency to "rip", forming vacuum or air pockets and rendering the xylem vessel inoperative.
Conductivity
Pure water is actually a good insulator (poor conductor), meaning that it does not conduct electricity well. Because water is such a good solvent, however, it often has some solute dissolved in it, most frequently salt. If water has such impurities, then it can conduct electricity much better, because impurities such as salt comprise free ions in aqueous solution by which an electric current can flow.
Electrolysis
Water can be split into its constituent elements, hydrogen and oxygen, by passing a current through it. This process is called electrolysis. Water molecules naturally disassociate into H+ and OH- ions, which are pulled toward the cathode and anode, respectively. At the cathode, two H+ ions pick up electrons and form H2 gas. At the anode, four OH- ions combine and release O2 gas, molecular water, and four electrons. The gases produced bubble to the surface, where they can be collected.
Reactivity
Chemically, water is amphoteric: able to act as an acid
or base. Occasionally the term hydroxic acid is
used when water acts as an acid in a chemical reaction.
At a pH of 7 (neutral), the concentration of hydroxide ions
(OH-) is equal to that of the hydronium (H3O+)
or hydrogen ions (H+) ions. If the equilibrium
is disturbed, the solution becomes acidic (higher concentration
of hydronium ions) or basic (higher concentration of hydroxide
ions).
Water can act as either an acid
or a base in reactions. According to the Brønsted-Lowry system, an acid is defined
as a species which donates a proton (an H+ ion) in a reaction, and a base as one
which receives a proton. When reacting with a stronger acid, water acts as a base;
when reacting with a weaker acid, it acts as an acid. For instance, it receives
an H+ ion from HCl in the equilibrium:
HCl + H2O ---> H3O+ + Cl-
Here water is acting as a base, by receiving an H+ ion. An acid donates an H+ ion, and water can also do this, such as in the reaction with ammonia, NH3:
pH in Practice
In theory, pure water has a pH of 7. In practice, pure water is very difficult to produce. Water left exposed to air for any length of time will rapidly dissolve carbon dioxide, forming a solution of carbonic acid, with a limiting pH of ~5.7 (reference: Kendall, J. (1916), Journal of the American Chemical Society 38 (11): 2460-2466).
Purifying water
Purified water is needed for many industrial applications, as well as for consumption. Humans require water that does not contain too much salt or other impurities. Common impurities include chemicals or harmful bacteria. Some solutes are acceptable and even desirable for perceived taste enhancement. Water that is suitable for drinking is termed potable water.
Six popular methods for purifying water are:
- Filtering: Water is passed through a sieve that catches small particles. The tighter the mesh of the sieve, the smaller the particles must be to pass through. Filtering is not sufficient to completely purify water, but it is often a necessary first step, since such particles can interfere with the more thorough purification methods.
- Boiling: Water is heated to its boiling point long enough to inactivate or kill microorganisms that normally live in water at room temperature. In areas where the water is "hard", (containing dissolved calcium salts), boiling decomposes the bicarbonate ion, resulting in some (but not all) of the dissolved calcium being precipitated in the form of calcium carbonate. This is the so-called "fur" that builds up on kettle elements etc. in hard water areas. With the exception of calcium, boiling does not remove solutes of higher boiling point than water, and in fact increases their concentration (due to some water being lost as vapour)
- Carbon filtering: Charcoal, a form of carbon with a high surface area due to its mode of preparation, adsorbs many compounds, including some toxic compounds. Water is passed through activated charcoal to remove such contaminants. This method is most commonly used in household water filters and fish tanks. Household filters for drinking water sometimes also contain silver, trace amounts of silver ions having a bactericidal effect.
- Distilling: Distillation involves boiling the water to produce water vapour. The water vapour then rises to a cooled surface where it can condense back into a liquid and be collected. Because the solutes are not normally vaporized, they remain in the boiling solution. Even distillation does not completely purify water, because of contaminants with similar boiling points and droplets of unvaporized liquid carried with the steam. However, 99.9% pure water can be obtained by distillation.
- Reverse osmosis: Mechanical pressure is applied to an impure solution to force pure water through a semi-permeable membrane. The term is reverse osmosis, because normal osmosis would result in pure water moving in the other direction to dilute the impurities. Reverse osmosis is theoretically the most thorough method of large-scale water purification available, although perfect semi-permable membranes are difficult to create. on exchange chromatography: In this case, water is passed through a charged resin column that has side chains that trap calcium, magnesium, and other heavy metal ions. In many laboratories, this method of purification has replaced distillation, as it provides a high volume of very pure water more quickly and with less energy use than other processes. Water purified in this way is called deionized water.
Wasting Water
Wasting water is the abuse of water, i.e. using it unnecessarily. An example is the use of water, particularly water purified to human safe drinking standards, in unnecessary irrigation. Also, in homes, water may be wasted if the toilet is flushed unnecessarily or the tank leaks. Causing water to become polluted may be the biggest single abuse of water. To the extent that a pollutant limits other uses of the water, it becomes a waste of the resource, regardless of benefits to the polluter.
Mythology
Water is one of the four classical elements along with fire, earth and air, and was regarded as the ylem, or basic stuff of the universe. Water was considered cold and moist. In the theory of the four bodily humours, water was asssociated with phlegm. Water was also one of the Five Elements in Chinese Taoism, along with earth, fire, wood, and metal.
Water rights and development
UNESCO's World Water Development Report (WWDR, 2003) from its World Water Assessment Program indicates that in the next 20 years the world is facing an unprecedented lack of drinking water. The quantity of water available to everyone is predicted to decrease by 30%. The causes are contamination, global warming and political problems. More than 2.2 million people died in 2000 from illnesses related to the consumption of contaminated water. In 2004, the UK charity WaterAid reported that a child dies every 15 seconds due to easily preventable water-related illnesses.The report indicates large global disparities in the raw volume of available water: from 10 m³ per person per year in Kuwait to 812.121 m³ in French Guiana. However, richer countries such as Kuwait can more easily cope with low water availability. In the United States water law is divided between two legal doctrines: riparian water rights, used in the eastern and southern states where there is an abundance of water and the appropriation doctrine (or Colorado doctrine) used in the arid western states.