What is the geometry of the methane molecule?
The simplest hydrocarbon , methane is a gas with a chemical formula of CH4 and a molecular weight of 16.04.
-------------->spin on -------->-
spin off
------>space fill/cpk
-------->stick
----> ball-and-stick
To Rotate the Molecule--->Left Click and Drag
To Zoom-->>Left Click + hold Shift button and Drag Vertically
Jsmol Menu --->>Right-Click
Try this!!
Change to wireframe first:
Click on right mouse button over box
Style --> Schemes --> wireframe
Label atoms:
Style -->Label ---> atom number
To measure angles: hold left mouse bottom over atom and double click on atom 1, drag to second atom (center atom) single click center atom, drag and double click atom 3.
Measure the following angles:
<415, <512, <213, <314
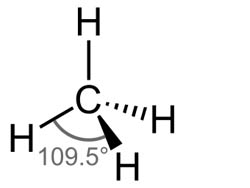
Below is shown the molecule Methane.
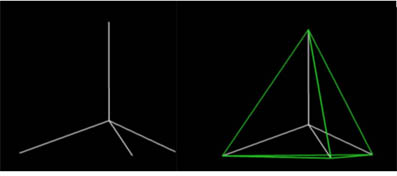
What polyhedral stucture would you think the methane molecule best represents? View the flash video below to see...

About Methane:
Methane is a principal component of natural gas. When a single molecule of methane is burned in the presence of oxygen releases one molecule of CO2[carbon dioxide) and two molecules of H2O (water).
CH4 + 2O2 ---> CO2 + 2H2O
The strength of the carbon hydrogen covalent bond in methane is among the strongest in all hydrocarbons.
Pure methane is odorless, but when used as a fuel it is usually mixed with small quantities of strongly-smelling sulfur compounds such as ethyl mercaptan to enable the detection of leaks.
Methane is a greenhouse gas with a global warming potential of 22 (meaning that it has 22 times the warming ability of carbon dioxide). Methane results from the decomposition of certain organic matters in the absence of oxygen. It is therefore also classified as a biogas.
Assessment -- Try these questions--
1) What is the measurement for any of the H-C-H angle to the closest tenth degree?
Please enter your answer in the space provided:
2) What is the measure of the C-H bond in methane?
3)How many faces does a tetrahedron have?
Explain it with Molecules
- Why is water such a good solvent?
- Why does ice float?
- Why do solids, liquids and gases behave differently?
- What is the geometry of methane?
- What's the difference between alpha and beta glucose?
- How does caffeine work in the brain?
- What is the difference between sucrose and fructose?
- Why is carbon monoxide so dangerous?
- Why is graphite so soft if it is made of only carbon?
- What is the difference between Carbyne and Graphite?
- Why is the fullerene and similar structures the cornerstone of nanotechnology?
- How big is a nanotube?
- Why does table salt have a cubic crystal shape?
- What is the structure of the benzene molecule?
- Why do carcinogens cause cancer?
- What causes Sickle Cell Anemia?
- What is the difference between sodiukm nitrite and nitrate?
- How do drugs work?
Molecules of Life Resources