Why does ice float?
Ice floats because it is less dense than water. Water has a density of 1.0 gm/cubic cm.
The density of ice Ih is 0.931 gm/cubic cm.
But, why is ice less dense than water if both are made up of molecules of H2O?
-------------->spin on -------->- spin off
------>space fill/cpk -------->stick ----> ball-and-stick
To Rotate the Molecule--->Left Click and Drag
To Zoom-->>Left Click + hold Shift button and Drag Vertically
Jsmol Menu --->>Right-Click
Try this!!
Change to wireframe first:
Click on right mouse button over box
Style --> Schemes --> wireframe
Label atoms:
Style -->Label ---> atom number
Measure the following angles:
<415
<512
<213
<314
To measure angles: hold left mouse bottom over atom and double click on atom 1, drag to second atom (center atom) single click center atom, drag and double click atom 3.
------>space fill/cpk -------->stick ----> ball-and-stick
Liquid water has a partially ordered structure in which hydrogen bonds are constantly being formed and breaking up.
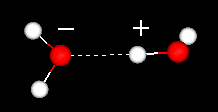
On the other hand ice has a rigid lattice structure.
In liquid water each molecule is hydrogen bonded to approximately 3.4 other water molecules.
In ice each each molecule is hydrogen bonded to 4 other molecules.
Compare the two structures below. Notice the empty spaces within the ice structure.
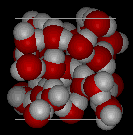 |
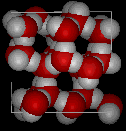 |
In ice Ih, each water forms four hydrogen bonds with O---O distances of 2.76 Angstroms to the nearest oxygen neighbor. Because of ordered structure in ice there are less H20 molecules in a given space of volume.
Try this --
Try this --
1) Measure the O-O distances between any two adjacent oxygen atoms in ice shown in the above structure..
Please enter your answer in the space provided:
2) Measure the O-O-O angle formed between adjacent oxygen atoms in ice.
Unlike ice -- water at room temperature is in constant motion -- hydrogen bonds are constantly formed and being broken-- click here to see water molecules in motion
Explain it with Molecules
- Why is water such a good solvent?
- Hydrogen bonds in water and ice
- Why does ice float?
- Why do solids, liquids and gases behave differently?
- What is the geometry of methane?
- What's the difference between alpha and beta glucose?
- How does caffeine work in the brain?
- How does soap work?
- What is the difference between sucrose and fructose?
- Why is carbon monoxide so dangerous?
- Why is graphite so soft if it is made of only carbon?
- What is the difference between Carbyne and Graphite?
- Why is the fullerene and similar structures the cornerstone of nanotechnology?
- How big is a nanotube?
- Why does table salt have a cubic crystal shape?
- What is the structure of the benzene molecule?
- Why do carcinogens cause cancer?
- What causes Sickle Cell Anemia?
- What is the difference between sodium nitrite and nitrate?
- How do drugs work?
Molecules of Life Resources