Mathematics and Molecules- A STEM Activity
What is the relationship between molecular modeling and mathematics?
This
activity uses Jsmol
_____________________________________
1 Dimension, 2 Dimensions, 3 Dimensions...
In this lesson we will take a look at the 3-Dimensional world we live in. Look at the image below. You will see two points.

A line has only one dimension --length. The line has no endpoints and extends forever in both directions. That is why when a line is drawn, we see arrows at both ends. On the other hand when we draw a line segment on a piece of paper it has endpoints.

Question 1: How many endpoints does a line segment have?
Questions 2: What is the figure below called?
The line, ray and line segment are all examples of 1-Dimensional objects since they only have length.
On the other hand a square has both length and height.
Question 3: How many dimensions does a square have?
A square or rectangle is really a plane, a 2-Dimensional object that has no thickness.
The Geometry of 2 Dimensions
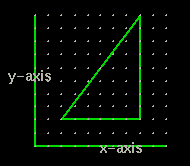
When we see a graph on a page we really don't think in terms of dimensions. But the graph above has only two axis or two dimensions. It is a plane.
 Click
on Graph to see that it only has 2-D
Click
on Graph to see that it only has 2-D
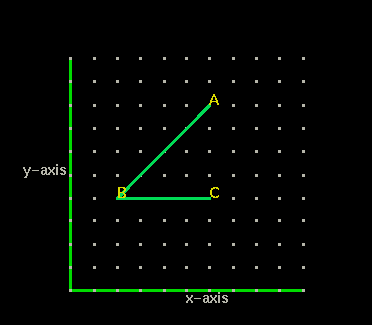
Point B has the coordinates (2,4). Starting from the origin (0,0) to reach point B we count 2 spaces to the right, then four spaces up.
Question 4: What are the coordinates of point A?
The
Geometry of 3 Dimensions
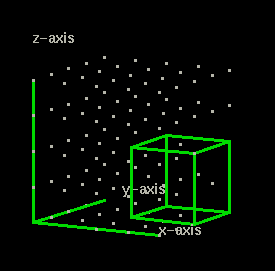
Compare the graph above (showing the cube) to the one shown in the above that contains angle ABC. What is different in this graph? Besides the x-axis and y-axis what do you see?
This graph has another axis, the z-axis. When we locate a point in three dimensions we need to use three coordinates. The origin in a two dimension plot is written (0,0).
Question 7: How do you write the coordinates for the origin in a 3-dimension plot?
What does all this have to do with molecules? Molecules are solid 3 dimensional objects in 3 dimensional space. To describe molecules accurately we must describe their postion in 3-D space.
-------------->spin on -------->-
spin off
------>space fill/cpk
-------->stick
----> ball-and-stick
To Rotate the Molecule--->Left Click and Drag
To Zoom-->>Left Click + hold Shift button and Drag Vertically
Jmol Menu --->>Right-Click
PDB File for Salt Molecule click here.
Look at the salt crystal structure above. Each atom is represented on the x,y,z coordinate axis as a single ball.
In the next section we will look at molecules and their relationship to mathematics.
GO TO PART II
Explain it with Molecules
- Why is water such a good solvent?
- Why does ice float?
- Why do solids, liquids and gases behave differently?
- What is the geometry of methane?
- What's the difference between alpha and beta glucose?
- How does caffeine work in the brain?
- How does soap work?
- What is the difference between sucrose and fructose?
- Why is carbon monoxide so dangerous?
- Why is graphite so soft if it is made of only carbon?
- What is the difference between Carbyne and Graphite?
- Why is the fullerene and similar structures the cornerstone of nanotechnology?
- How big is a nanotube?
- Why does table salt have a cubic crystal shape?
- What is the structure of the benzene molecule?
- Why do carcinogens cause cancer?
- What causes Sickle Cell Anemia?
- What is the difference between sodium nitrite and nitrate?
- How do drugs work?
Molecules of Life Resources